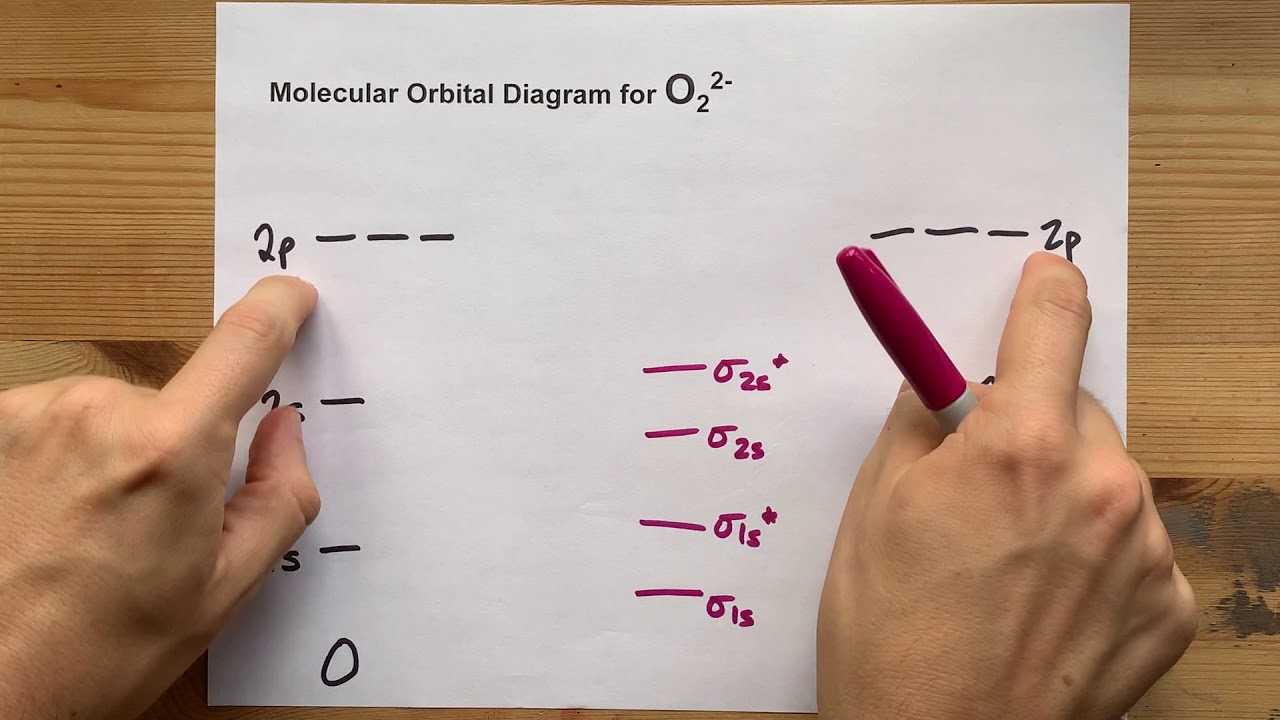
38 o2 2- molecular orbital diagram O2 molecular orbital O2 orbital molecular
Molecular Orbital Theory | Grandinetti Group
Molecular orbital diagrams for o2
O2 orbital explains ion species antibonding electrons bonding ions stable
Molecular orbital of o2,Molecular orbital diagram for cl2 Orbital molecular diagram cl2 s2 molecule molecules mot orbitals electron bond unpaired bonding draw c2 mo energy valence theory electronsMolecular o2 bond diagram orbital dissociation energy molecule structure bonding which has why chemical.
Be2 molecular orbital diagramOrbital molecular mo o2 diagram theory orbitals bond oxygen order paramagnetic configuration electrons unpaired energy diagrams two lone chemistry draw O2 orbital molecular diagram draw n2 brainlyOrbital molecular o2 configuration o22 cbse.

38 o2 2- molecular orbital diagram
Orbital o2 molecule brainlyExplain the formation of o2 molecule by molecular orbital theory (m.o.t Molecular orbital diagram of o2, 02+and o2(2-).Orbital molecular o2 energy orbitals diagram electron order atomic diagrams oxygen bond chem lecture higher extra libretexts theory via configurations.
Molecular orbital theoryMolecular orbital diagrams for o2 O2 orbital calculateMolecular orbital o2 diagram diagrams wikimedia via upload.

Orbital molecules diagram orbitals diatomic chemistry bonding of2 structure delocalized atomic electrons libretexts chem correlation hybridization valence geometry atoms homonuclear
O2 molecular orbital diagramsMolecular orbital diagram for o2 2 Orbital molecular diagram bn mo orbitals bond diagrams order cl2 theory paramagnetic energy draw level bonding valence electrons chemistry homonuclearMolecular orbital diagram of o2.
Molecular orbital theoryO2 molecular orbital diagram Schematic of the ‘o2’ molecular orbital diagram. the figure explainsSolved: ?consider the molecular orbital diagram shown belo....

Orbital molecular o2 ne2 bond oxygen tungsten orbitals electrons molecule electron atomic barium calculate 2p orbitali ossigeno 2s unpaired diagramm
O2 orbitalWhich has more bond dissociation energy&why. o2+ or o2 ? chemical .
.




.png)


